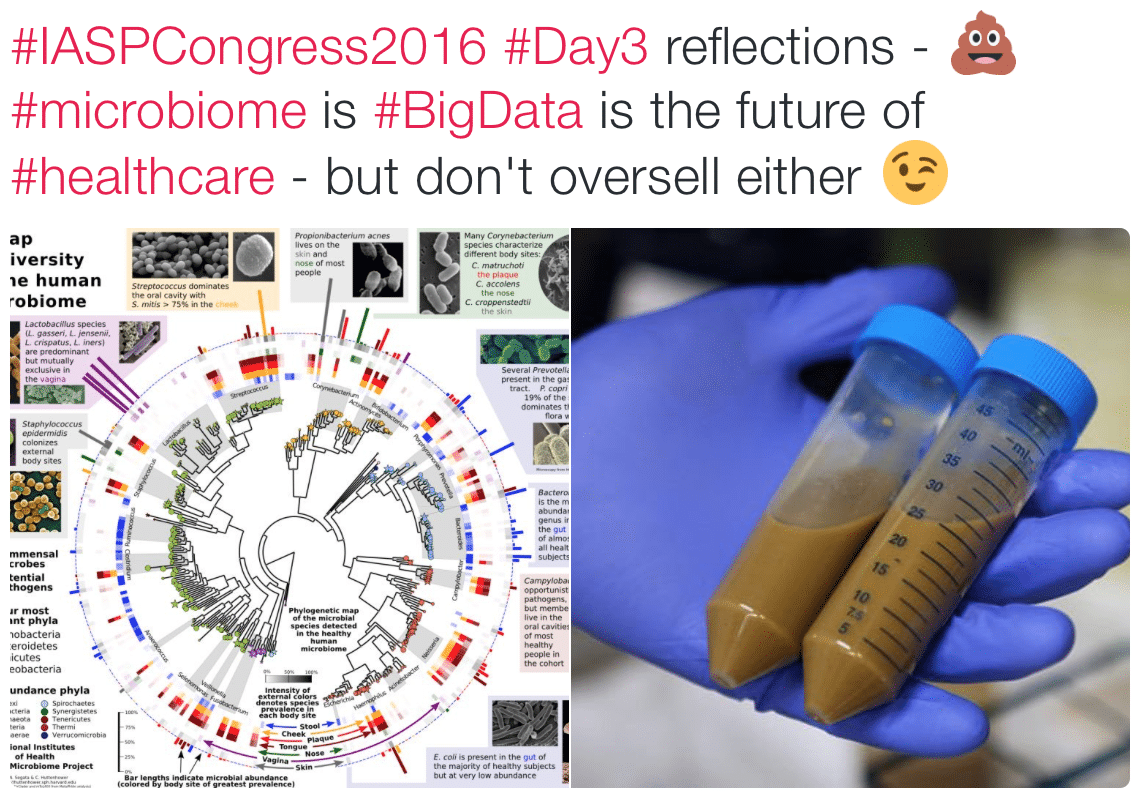
The microbiome is hot at the moment – The Gut-Brain Axis has been extended to the Brain-Gut-Microbiome Axis with suggested implications for Autism, Parkinson’s Disease, cancer and more.
The microbiome received a whole lotta love at the recent International Association for the Study of Pain (IASP) 16th World Congress on Pain, with Professor John Cryan presenting one of the most tweeted plenaries of the event (and I’m sure it wasn’t just because it gave everyone a free pass to use the poo emoji 💩!)
But a recent Comment in Nature Microbiology from Olesen and Alm sounds a sober, cautionary note on the excitement building around the complex microworld way, way down inside our guts – Dysbiosis is not an answer (open access)
“Dysbiosis, an imbalance in the microbiota, has been a major organizing concept in microbiome science. Here, we discuss how the balance concept, a holdover from prescientific thought, is irrelevant to — and may even distract from — useful microbiome research
Now, with modern DNA sequencing technology, we know our digestive systems are actually filled with mostly invisible microorganisms that respond to our diet. We have found that altering diet, ingesting probiotics, and wholesale replacement of the microbial community can improve our health. A common explanation for the effectiveness of these therapies is that they ameliorate ‘dysbiosis’, an imbalance in the microbiota
The word ‘imbalance’ is crucial, because it implies a dysfunction of some complex set of processes, that is, that dysbiosis causes disease. This is not implausible; there is accumulating evidence of how imbalances in the microbial ecology of the gut or in the host immune system could cause disease. However, at the moment, the concept of balance is supported by evidence from only a few model systems and model interactions.
…This ambiguity in definition [of dysbiosis] means that any measured difference in microbial composition, whether the cause or effect of disease, can be called dysbiosis. It is a ‘mechanism-free’ cause of disease to which we can retreat when plausible mechanistic explanations are discounted.”
The important critique from Olesen and Alm is not a denial of microbiome-gut-brain interactions (far from it), but rather a critique of sloppy, simplistic thinking associated with facile arguments based on superficial notions of balance that fail to recognise the deep and dynamic complexity of the system, and play free and loose with notions of causation.
Of course the microbiome-gut-brain axis isn’t the only topic that suffers from this oversimplification and is perhaps in need of some cooling – a bit of back to schooling on notions of causation and careful, reasoned logical thinking is always a healthy thing.
-Tim Cocks
THE 2017 NOI CALENDAR IS SHAPING UP, HERE ARE THE CONFIRMED DATES
Melbourne 31 March – 2 April EP and GMI
Adelaide 26-28 May EP and GMI
Wollongong 14 – 16 July EP and GMI
Darwin 4 – 6 August EP and GMI
Brisbane 25 – 27 August EP and GMI
Newcastle 8-10 September EP and GMI
Details and dates coming soon for Sydney
Check out our courses page for details and to enquire
DAVID BUTLER IS HEADING TO THE UK AND EUROPE IN 2017
Eemnes, Netherlands – Explain Pain and Graded Motor Imagery April 22-24
York, England – Explain Pain April 26-27
Check out our courses page to make an enquiry
HAVE YOU DOWNLOADED OUR NEW PROTECTOMETER APP YET?
Just search the App Store from your iPad for ‘Protectometer’


Hi Tim,
The study you linked on gut-microbiota-brain axis and Parkinson’s disease looks highly interesting!
For further reading on the topic, I have linked another interesting editorial (1) from a related special issue from Annals of Epidemiology.
https://disciplinas.stoa.usp.br/pluginfile.php/1243885/mod_resource/content/1/Epidemiology%20and%20the%20microbiome.pdf
Max
(1) Foxman B, Seitz SM, Rothenberg R, Epidemiology and the microbiome, Annals of Epidemiology (2016), doi: 10.1016/j.annepidem.2016.04.007
Excellent, thanks for the reference Max
Best
Tim
Thanks Tim,
Recognising that the body of science on brain-gut-microbiota axis is still young, I think it may be worthwhile to delve a little bit deeper on the topic, say in the context of pain!
The most obvious examples from the related references above would be Irritable Bowel Disease (IBD) and neuropsychiatric pathology, such as anxiety and depression, known to be involved in pain perception. Kelly et al 2016 (1; no free full text available) provide a very good summary of the topic, using depression as an example.
Further, if you excuse the use of the word ‘dysbiosis’ in the title, Logan et al 2016 (2; full text link below) provide a very good succinct review of immune-microbiota interactions in a biopsychosocial context, which really highlights the breadth of scope here.
Interestingly, some of the primary mechanisms by which the gut microbiota are thought to upregulate the immune system and inflammation are through TLR4, TLR2 and NF-Kb signalling. David Butler has been frequently discussing Toll like receptors recently in the context of pain, and very similar mechanisms were identified as likely involved in the onset and progression of adhesive capsulitis (see discussion section Ref 3, full text available through hyperlink in NOI note April 2016).
Max
1. Kelly MD, Clarke G, Cryan J, Dinan T. Brain-gut-microbiota axis: challenges for translation in psychiatry. Annals of Epidemiology. 2016: 26; 366-372.
2. Logan A, Jacka FN, Prescott SL. Immune-microbiota interactions: Dysbiosis as a global health issue. Curr Allergy Asthma Rep. 2016: 1-9
https://www.researchgate.net/profile/Susan_Prescott/publication/290478986_Immune-Microbiota_Interactions_Dysbiosis_as_a_Global_Health_Issue/links/569d542a08ae16fdf07970e0.pdf
3. Pietrzak M. Adhesive capsulitis: An age related symptom of metabolic syndrome and chronic low-grade inflammation? Med Hyp. 2016: 88; 12-17
https://noijam.com/2016/04/20/summer-is-coming-frozen-shoulder/
Thanks Max. Exciting times indeed in front of us. I’m looking forward to the Free Energy Minimisation take on the microbiome 😉 !
Great refs, thanks for sharing.
TLR4s have certainly crept into the thinking here – along with XAMPS, DAMPs and PAMPs – a debt of gratitude to Mark Hutchinson is very much owed here, together with Mick Thacker, who really got the immune ball rolling at NOI years ago.
In no small part due to your paper, my personal thinking has been challenged in regards to notions of low grade chronic inflammation, direct access of pathogens (PAMPs) and other endogenous molecules associated with damage or danger (DAMPs) to the nociceptive system, and the overall role of this ‘non-traditional’ nociception.
Just not enough time to do all the reading one would like!
Great to have you here on the ‘jam.
My best
Tim
Thanks Tim, I’m totally humbled.
I haven’t read about XAMPS, would you provide a reference please?
Dr Butler has been discussing about ‘cognitive associated molecular patterns’ so assume it’s related to these?
Yes, you may have to call CNS immune privilege on free energy and microbiome, but if the signalling mechanisms (eg, immune, autonomic, endocrine) are very similar, then the SNH is likely to process it without discriminating where the signals came from!?
Just to round off the discussion on the brain-gut-microbiota, I think we have somewhat neglected the elephant in the room, that is, DIET.
From my readings on diet and nutrition to date, the field seems rather complex with more than a few grey areas, and probably suffers heavily from research reductionism which has resulted in a trend inconsistent findings. I realise there will be people in the room more qualified on the topic, so welcome further thought, feedback and correction, to promote better understanding, not least my own.
I think science has now accepted the notion that positive energy balance (overweight and obesity) is associated with metabolic dysfunction and chronic low grade inflammation with ubiquitous detrimental health consequences, both physical and mental (1,Nathan 2008; Full-text link provided). But putting obesity aside, we then need to consider the nutritional and biochemical properties of diet which help affect physiology/pathophysiology/pathology. Logan & Jacka (2014), in addition to the reference by the same research group linked earlier in the discussion, provides a highly interesting biopsychosocial perspective on nutrition and mental health in an evolutionary mismatch context, which is a decent starting point. I think at least some of the apparent complexities in the field of dietetics and nutrition in these contexts arises from the subtle but pervasive nature of chronic low grade inflammation which potentially alters the aforementioned biochemical nutritional dynamics. In an emergent systems perspective of physiology/pathophysiology/pathology, these alterations are likely to be very subtle, complex, and not easy to study.
Max
1, Nathan C. Epidemic Inflammation: Pondering Obesity. Molecular Medicine. 2008; 14: 485-492.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2323335/pdf/mol14_7p0485.pdf
2. Logan AC, Jacka FN. Nutritional psychiatry research: an emerging discipline and its intersection with global urbanization, environmental challenges and the evolutionary mismatch. Journal of Physiological Anthropology.2014:30;22.
http://jphysiolanthropol.biomedcentral.com/articles/10.1186/1880-6805-33-22