There’s a cautionary tale in the story of radiological investigations for ‘musculoskeletal’ pain. As imaging improved and the pictures got clearer and shinier, those referring for scans, reporting on scans and interpreting scans for patients, grew more and more confident that they could visualise the ’cause’ (or lack thereof) of an individual’s pain experience. This tumescent confidence has not been without its problems, and every therapist will have first hand experience of a person whose pain has been derided and denied as a result of “nothing showing up on the scan”, or equally, the patient who falls off the precipice after 6 weeks of steady progress because their long awaited MRI demonstrated a ‘bulging disc’.
There are attempts now to put out the dumpster fire that imaging for low back pain has become, such as the Choosing Wisely initiative and research into the possible consequences of the way radiology reports are written.
But it seems a different chapter of the radiology-for-pain guild haven’t got the memo and are possibly hurtling down the track towards problems.
Of bulges and blobs
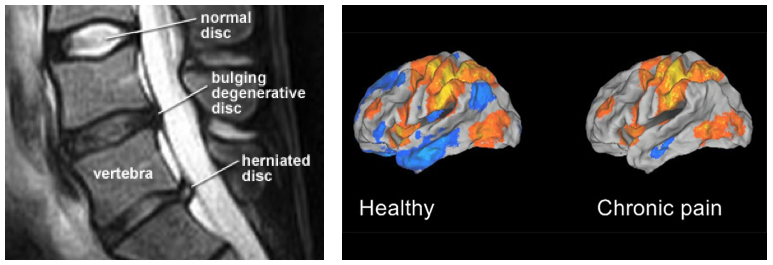
In a review article in the current Clinical Journal of Pain, Evaluation of Chronic Pain Using Magnetic Resonance (MR) Neuroimaging Approaches, What the Clinician Needs to Know (Kumbhare et al 2017), the authors suggest
“…most clinicians are not aware of the capabilities of advanced MRI methods in assessing cortical manifestations of chronic pain. In addition, many clinicians are not aware of the cortical alterations present in individuals with chronic pain.“
The basic idea (well intentioned I’m sure) is to begin to set out a path for clinicians to use fMRI imaging of the brain to help diagnose, track and treat pain.
But in the same issue Karen Davis, the great debunker of fMRI and pain overstatement, and David Seminowicz have written a commentary on the Khumbhare et al (2017) paper; Insights for Clinicians From Brain Imaging Studies of Pain (Davis and Seminowicz 2017) noting some fundamental issues with the idea
Neuroimaging has taught us a lot about the brain in chronic pain, but individual variability means that these findings cannot be generalized across conditions
The first point to take away from the Kumbhare et al review is that there were no specific abnormalities in common across the studies, likely owing to the variability between studies, including differences in chronic pain condition, age, sex, and co-occurring symptoms and diseases.
The effect of sex differences is not always often fully considered in brain imaging studies despite chronic pain conditions being more prevalent in women (eg, fibromyalgia, irritable bowel syndrome, temporomandibular disorder) or more prevalent in men (eg, back pain, anklyosing spondylitis).
Thus, it is important for a clinician to know that there is vast intersubject variability (including sex differences) in the general response to and coping with painful stimuli, brain circuitry, and connectivity.
Can brain biomarkers replace pain ratings?
Kumbhare et al state that “compared with self-reporting approaches, objective imaging techniques are expected to potentially lead to better pain management.” We find this statement problematic for several reasons. We argue that pain ratings are the best outcome measure we have for treating chronic pain and will likely always remain that way – imagine for example a patient who’s brain is deemed “recovered” based on our neuroimaging techniques, but still reports suffering intolerable pain most of the day. (emphasis added)
The last comment above (in bold) is the telling one.
-Tim Cocks
Melbourne, 31 March – 2 April EP + GMI SOLD OUT
Adelaide, 26-28 May EP + GMI
Wollongong, 14-16 July EP + GMI
Darwin, 4-6 August EP + GMI
Brisbane, 25-27 August EP + GMI
Newcastle, 8-10 September EP + GMI



Well said TC!
Clever choice about the dumpster fire as well… unless immediately next to a structure (or dry bush) usually fairly benign. Sure they can be hot, smoky and noxious… but usually pretty much self contained and will fizzle out without too much worry. Maybe like many of the findings on a typical MRI… fairly well contained and frequently benign.
cheers – Steve
Hi Steve
Interestingly, as I was looking for a good dumpster fire pic, I read a little about them, as well as the history of the ‘meme’. It seems that dumpster fires can be incredibly dangerous and toxic and firefighters always approach them with great care – a bit like we should with MRI for a majority of ‘MSK’ conditions perhaps!
Thanks for stopping by
Tim
Tim, A timely post and well written, especially since a very popular blog titled “Imaging” recently. What if we acknowledged that “pain is an output” written of many times? It is, however, invisible.
Barrett
Hi Barrett
For me, that notion of invisibility is key – I’m not confident that we will ever be able to “see” the experience of pain in any scan (at least in my lifetime).
My best
Tim
Tim, By the way, I wrote the aforementioned blog post.
Barrett
Thanks Barrett, i had entertained that notion.
Tim
Hi Tim,
Do you think we should draw a distinction between chronic and acute musculoskeletal pain episodes and the use of MRI, given the strong title of the thread? I believe they are very commonly utilised in elite sport. Or should clinical examination and reasoning without imaging sufficient in all cases?
A couple of interesting examples from my near 20 years practice, granted these are quite rare.
1) The only time I have had to call an ambulance for a client in private practice,(severe LBP, sciatica, potential cauda equina syndrome), as it looked the only option for the client to be able to make it out of the clinic, the MRI result was NAD (not even odema!).
2) A gentleman approximately 60yo who rode his bike daily a few miles got a flat tyre and decided to use his granddaughter’s bike instead (much smaller bike). Interestingly there was no pain during the ride, it started almost immediately after, but I was seeing him in the clinic approximately 12months later at the back end of the constant sciatica episode since the event, but his main problem was a potentially permanent partial foot drop. I can’t remember what imaging he had, but wonder if an MRI could be relevant in such a context.
Max
Thanks Max
I think MRIs are fantastic, an amazing technology and I wouldn’t be without them. But, I think there is ample evidence now to state with confidence that a majority of ‘findings’ have no correlation to an individual’s experience of pain. However the conflation of findings with experience has led to the situation (the dumpster fire) where the victim of the MRI is either disbelieved, or experiences additional iatrogenic suffering.
I think a lot of clinical reasoning has been ‘offloaded’ to scans, rather than scans being a possible (and valid if the presentation supports it) outcome of the CR process.
Great examples, I have a memory of a patient similar to your eg 1 – but deep thoracic pain, difficulty breathing, generally feeling very poor. I took one look at her in the waiting room (sweaty, a bit grey…) and called the ambulance. Nada on all the scans!
For eg 2, I would ask, which bit of this gentleman would you MRI!
Thanks for taking the time to drop by
My best
Tim
Having just about reached 40 years of clinical practice, I utterly agree with your comment that a lot of clinical reasoning is off-loaded into scans. It is a sad state of affairs because I have always found the true joy of my clinical practice is to find as accurate a diagnosis as I can with appropriate, therefore more likely successful treatment and management. Reliance on imaging prevents development of clinical diagnostic expertise. I wonder if this is instrumental in the loss of good physios from the profession?
Kath
Kathy (and, of course, Tim),
My fascination with the first season of “House” had largely to do with my fascination with him as a diagnostician. In my area, in the skilled nursing facilities, I have described PTs as “a warm body with a license.” This got a boost on my blog and I’ve seen nothing to change this. Diagnosis is driven by tradition, advertisement, and expectation. Money (reimbursement) raises its head here, though few want to talk of this. What makes me smile is that the “doctorates” in the US think that they’re be asked to “think.” It’s not likely.
I’m a cynic, I know.
Barrett
Wholeheartedly agree Tim re: pain ratings as best outcome measure for treating chronic pain!